Pharmaceutical And Medical Device Primary Packaging And Circular Economy

The volume of packaging waste globally is over 500 million tonnes, of which around 80 million tonnes is generated in the European Union and a similar volume in the USA. Despite significant advances in recycling, consumer education, and packaging design, the growth in packaging consumption continues to outstrip recycling capacity and technological advances.
Additionally, although the total volume of packaging waste is stabilizing in many OECD countries, this is driven by reduced paper, cardboard, and glass packaging. Significantly, plastic packaging waste continues to increase, representing almost 150 mio tonnes annually. In the USA, only 14% of this is recycled; in the EU, the data is better at 38%. Globally, 22 mio tonnes of plastics leak into the environment yearly, ending up in our rivers, lakes, seas, and forests.

As the producer of 4.6% of global greenhouse gas emissions, the Healthcare industry is pressured to reduce its environmental impact. While consumer products have moved heavily away from single-use plastics, the healthcare industries have clung on, with the UK’s National Health Service being the largest single-use plastics user in Europe. Although Pharmaceutical and Medical Device packaging waste represents only around 1% of overall packaging waste, the industry mindset is that it cannot be recycled, and almost all ends up in landfills or incinerated.
Importance of Plastics in Packaging
Plastics play a vital and unavoidable role in the primary packaging of pharmaceutical products and medical devices. They are critical to the protection and shelf life of the content by providing a barrier to microbes, moisture, oxygen, and light. Ensuring products are in perfect condition after two years and the packages open without damage or contamination are much higher demands than most consumer goods packaging.
Adding in the critical need for correct labeling, child resistance, and the potential of counterfeiting, the regulatory environment of both Pharmaceutical and Medical Device packaging is extremely risk-averse and restrictive, making the change of packaging a costly and time-consuming activity.
The most common plastics in healthcare packaging are PE, PVC, PET, and PP, providing excellent protection and low interaction with sensitive content. However, especially in flexible packaging, these plastics are mostly combined with other polymers or aluminium to enhance the barrier properties, creating multi-material laminates that are near-impossible to recycle.
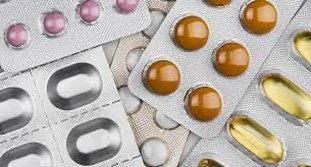
Blister packaging is the most popular form for tablets worldwide, representing around 60% of all doses, and PVC is used in over 90% of blister packs, usually with an aluminium foil lid.
Glass vials, syringes, and bottles are still heavily used, especially for vaccines. However, these have extremely high costs of production and recycling, as well as breakage risk, and are slowly being replaced by plastic alternatives. Plastic bottles, more commonly used in the US, are more frequently made of single polymers, particularly PET and PE, theoretically allowing them to be sorted into one of the plastic recycling streams.
Medical device primary packaging more often uses PET, PE, and PP, again, frequently combined with paper and barrier materials such as oPA and EVOH. The plastics in medical device packaging have the additional functionality of allowing the sterilization process while maintaining a microbial barrier post-sterilization.
With advances in paper production, specially reinforced and coated or impregnated paper can now provide a greater barrier and mechanical protection and can be considered viable alternatives for some Pharmaceutical and Medical Device products.
With this heavy reliance on multi-material plastic-based packaging and a cautious regulatory environment, it is unsurprising that Pharmaceutical and Medical Device companies are reluctant to commit to a major change.
The Packaging Industry as Key Enabler
For over 20 years, packaging companies have provided more sustainable options to standard packaging, downgauging foil, and film thickness, optimizing design to reduce pack size and components, and offering PP or PET as an alternative to PVC. However, the basic packaging has not changed for 40 years, and only in the past five years has the pace of innovation increased.
Large packaging companies such as Amcor made pledges to ensure all packaging is recyclable by 2025, and this has since been adopted across the healthcare packaging industry. This commitment was necessary to drive the sustainability agenda forwards, moving from an incremental to a transformational mindset and collaborating with global organizations such as Ellen MacArthur Foundation and the Healthcare Plastics Recycling Council.
The major primary packaging manufacturers have taken huge strides towards mono-polymer packaging, moving away from PVC in favor of PET, PP, and PE. These innovations have been enabled by investment in both R&D resources and advanced converting technologies.
With the broader availability of recyclable solutions, the major pharmaceutical companies have shown a greater willingness to trial these new packs, particularly for the Consumer Health and OTC markets, where end-users and retailers have a greater influence in the decision-making and the pressure to show eco-friendly credentials is greatest.
The largest PVC blister base film producer, Klockner Pentaplast, was the first to launch a mono polymer PVC-free blister in 2021 with kpnext™, a PET thermoformable base film, as an alternative to low-barrier PVC blisters, targeting the Consumer Health and OTC segments.

Since then, Amcor has launched an all-PE blister called Amsky™, which achieves a medium moisture barrier and allows use for a broader range of medications, including prescription drugs. This has been followed by Liveo Research, Tekni-Plex, Südpack Medica, and Etimex with their all-PET and all-PP blisters. These also promise medium barriers, with higher barrier options in development. The major PVC producers have been teaming up with film producers such as Huhtamaki and Adapa to provide lidding films, recognizing that collaboration is required to deliver results.
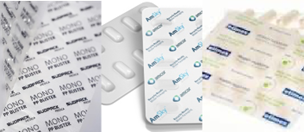
Medical device packaging has shown similar advances, as the producers aim to reduce the non-recyclable content to an absolute minimum, particularly limiting difficult-to-recycle materials such as oPA and PVC.
Some challenges remain to be solved in the higher barrier ranges to remove aluminium laminates and harmful vinyl chlorides and fluoropolymers from pharmaceutical blisters:
- Higher barriers provided by PVdC and PCTFE, representing around 15% of blister packs, have not yet been achieved with monopolymer blisters
- Aluminium cold-form blisters represent the highest barriers to moisture, oxygen, and light, and are around 20% of all blister packs, often prescription medications. While these can be recycled in the aluminium stream, this is only achieved by grinding and incinerating to recover the aluminium, and it is expensive, with limited results
The key messages here are that the packaging industry is starting to deliver the goods, providing Pharmaceutical and Medical Device companies with a range of recyclable alternatives, and they are prepared to work together to achieve this. So how quickly before a change is visible?
Significant Hurdles Ahead
Recyclable packaging solutions now available cover something like three-quarters of Pharmaceutical and Medical Device applications, and trialing at Pharma and Medical Device companies is gathering pace, particularly for new product launches. However, even if these packages are replaced in the coming few years, achieving a circular economy still needs to overcome major challenges. For example:
- Regulations today prevent Post-Consumer Recycled (PCR) content from being used in primary pharmaceutical and medical packaging. Patient safety demands that there can be no risk of contamination entering the value chain, and current mechanical recycling processes cannot guarantee this
- Contamination of Medical and Pharmaceutical packaging with biological, chemical, or toxic materials, even in empty packages, is considered too risky. For this reason, finished packaging is usually incinerated or simply goes into the trash and landfill, despite over 80% of healthcare waste being classified as non-risk
- Collection and separation of used packaging are underdeveloped, particularly for home-used medications and devices. A used pack may contain some residual medical or pharmaceutical content; currently, these are not accepted for recycling. The advice generally is to put used healthcare packaging in the trash. Some schemes have started to collect empty blister packs, such as Terracycle in Australia and Superdrug in the UK

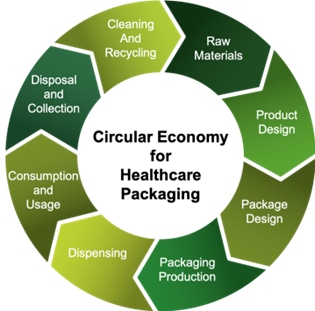
Moving to a Circular Economy
As the packaging producers have shown, collaboration along the value chain is needed to achieve circularity in healthcare packaging.
First and foremost, the Pharmaceutical and Medical Device Manufacturers must advocate for change. In addition to driving the change to sustainable and recyclable raw materials, the leading companies should drive the need to change regulations to make it easier for recycled plastics to enter the pharmaceutical packaging value chain safely.
The product design can ensure the optimum use of both product and packaging. For example, coated tablets leave less residuehttps://kradminasset.s3.ap-south-1.amazonaws.com/ExpertViews/Colinpic8.png on the packaging, making separation and cleaning much easier. Designing medical devices, such as drug delivery and diagnostic devices, to be multi-use will significantly reduce the amount of packaging needed.
The package design should ensure the efficient use of materials, with the Pharma and Medical Device companies collaborating early and closely with the packaging producers. Alternatives to plastic-based flexible packaging, such as the greater use of paper, rigid plastic bottles, and trays, can make a recyclable mono-material pack easier to realize.
In addition to the primary packaging, consideration needs to be given to secondary packaging, such as leaflets, as these can be a great source of waste, which could be replaced by electronic information on the primary packaging and secondary cartons.
Continued investment in packaging production technologies is required to enable further innovation toward mono-material packs across the range of Pharmaceutical and Medical Device packaging formats.
When dispensing or supplying the product, ensure the correct quantities, avoid over-supply, and educate the consumer about how to use the products and dispose of packaging correctly.
Ensuring correct usage requires continued education and communication.
The packaging design can help simplify usage and is also a critical carrier of information. Smart packaging and digitally printed data can provide users with easy-to-understand information to minimize waste and improve safety. Standardization of medical devices will also improve patient compliance.
The weakest link in the chain currently is the disposal and collection of empty packs. Currently, no widespread schemes allow users to return empty or part-empty packs. Unused pharmaceutical products are often flushed into the water system, contaminating the environment. Here, a collaboration between the healthcare industry, local and regional governments, waste processing companies, and regulators are required to allow unused medicines and devices to be collected, separated, cleaned, and for the packaging to enter the recycling streams.
Finally, further advances are needed in mechanical and chemical recycling processes to make the recycling of PET and polyolefins more widespread and cost-effective. Processes such as hydrolysis, pyrolysis, and gasification are well-established in the depolymerization of plastics, allowing their return to chemical production.
Summary
Primary healthcare packaging producers have made great strides in developing single-material packaging, especially mono polymer plastic packaging. Add to that the common use of rigid plastic bottles and trays and the advances in paper technologies, and there are a few reasons not to adopt recyclable packaging for most healthcare applications.
Before technology allows all healthcare packaging to be fully recycled, there is some way to go, which requires investment throughout the value chain. It cannot be left to the packaging companies alone to carry the cost of innovation.
To move from “recycle-ready” to “recycled” and a genuine circular economy, Pharmaceutical and Medical Device companies, regulators, and governments must align and proactively advocate for changes in the regulatory environment and enhanced collection and recycling.
This article was contributed by our expert Colin Reed
Frequently Asked Questions Answered by Colin Reed
1. How can we increase the recycling rate of plastic packaging waste, particularly in countries with less advanced recycling infrastructure?
The education of users and consumers is critical. Packaging can play a key role by being simple to use and containing easy-to-understand instructions that can be accessed without effort.
Secondly, improved collection processes to allow finished medicine and device packs to be disposed of correctly.
2. What are the most effective strategies for reducing the amount of single-use plastic in healthcare packaging while maintaining the necessary levels of protection and safety?
Medical devices should be designed to be multi-use where possible and to use fewer components. If the device contains chemical or toxic content, consider how this can be kept separate from the main device components, perhaps by using replaceable cartridges.
Much single-use plastic is unavoidable when considering items like catheters and tubing, so the correct collection and recycling of these is important. Correctly classifying plastics into risk categories means they can better be sorted so that the small amount of high-risk waste is not mixed with reusable or recyclable waste.
3. What are the most promising new materials and technologies for sustainable packaging, and how can they be scaled up and integrated into existing supply chains?
Current recycling technologies suggest that PE is the best plastic polymer for advanced recycling, as it is used across all industries, is most widely available, and can be most easily recycled. PET recycling is more problematic, as is the raw material supply chain for healthcare grades, and PP recycling is not yet as advanced. Ideally, advanced recycling of mixed polyolefins creates the most flexible option for healthcare packaging.
Paper technology is progressing rapidly and is making significant strides at providing as much functional barrier and protection as plastics across many applications. Even in pharmaceutical packaging, a paper blister or pouch can provide sufficient protection for various OTC and Consumer Health applications.
The most advanced plastic packaging material structures are being produced with cast coextrusion technologies, as these allow multiple layers of thin films, adding a high level of functional barrier films while using significantly less material. Advanced coating technologies, even developments using nanocoatings – coatings of <100nm – are showing promise.
Supply chains for polyolefins and paper are well-established, and the raw material suppliers can produce specific grades for healthcare. Production capacity for these converting technologies is ramping up as the larger packaging producers have invested heavily in the past five years. The greatest bottleneck seems to be in trialing and approving the new specifications, which requires significant time and effort in QA departments and testing laboratories.
4. How can we encourage consumers and businesses to prioritize sustainable packaging options and make it economically viable for companies to invest in eco-friendly solutions?
The first priority remains communication and education – ensuring that individuals and corporations know what it means to do the right thing. Reward schemes are worth considering when returning used packaging to pharmacies, clinics, doctors, hospitals, and retailers to further encourage consumers.
Health insurers, governments, healthcare companies, and retailers should sponsor these schemes, as they all benefit from increased recycling.
A further incentive is to penalize using non-recyclable packaging when a recyclable option is available. Some retailers are already threatening to de-list products in PVC packaging, but this is only realistic if there are alternatives. It cannot be solely the burden of Pharma and Medical Devices companies to pay for the retesting and re-registration of the packaging – the other organizations in the chain must support this investment.
Comments
No comments yet. Be the first to comment!
